
correspond to l = 0, 1, 2, 3, 4, 5…, respectively. These are labeled by the quantum numbers n, l, and j, where n is the radial quantum number, that is, the number of nodes (plus 1) in the radial wave function, l is the orbital angular momentum, and j is the total angular momentum due to the addition of the orbital and spin angular momenta. Typical single-particle levels of nucleons in a spherical mean-field potential. These magic numbers have been explained using a finite depth potential with a strong spin–orbit interaction, as is shown in Fig. For stable nuclei, it has been known that the shell closures occur when the number of neutron or proton takes the so called magic numbers given by 2, 8, 20, 28, 50, 82, and 126. Such shell structure in atomic nuclei have been evidenced by many phenomena, such as an increased binding energy, discontinuities of the nuclear radius as well as neutron and proton separation energies (corresponding to the ionization energy in atoms), and an increased excitation energy of the first excited state, all of which occur at the shell closures. In this picture, nucleons occupy single-particle orbits, like electrons in atoms, which naturally leads to the concept of shell structure and shell closures. It has been known that many properties of atomic nuclei can be understood in a simple mean-field approximation, in which nucleons move independently from each other in a common potential, that is, a mean-field potential generated as a consequence of the interaction among nucleons (see e.g., Bohr and Mottelson 1969 Ring and Schuck 1980). They consist of a small number of protons and neutrons, collectively called nucleons. Send us feedback about these examples.Atomic nuclei are located at the center of atoms and carry almost all the fraction of the mass of atoms. These examples are programmatically compiled from various online sources to illustrate current usage of the word 'periodic table.' Any opinions expressed in the examples do not represent those of Merriam-Webster or its editors. 2023 This year’s questions were as follows: What will be the 119th element on the periodic table? - Jared Beasley, New York Times, 18 Mar. Manasee Wagh, Popular Mechanics, 24 Mar. 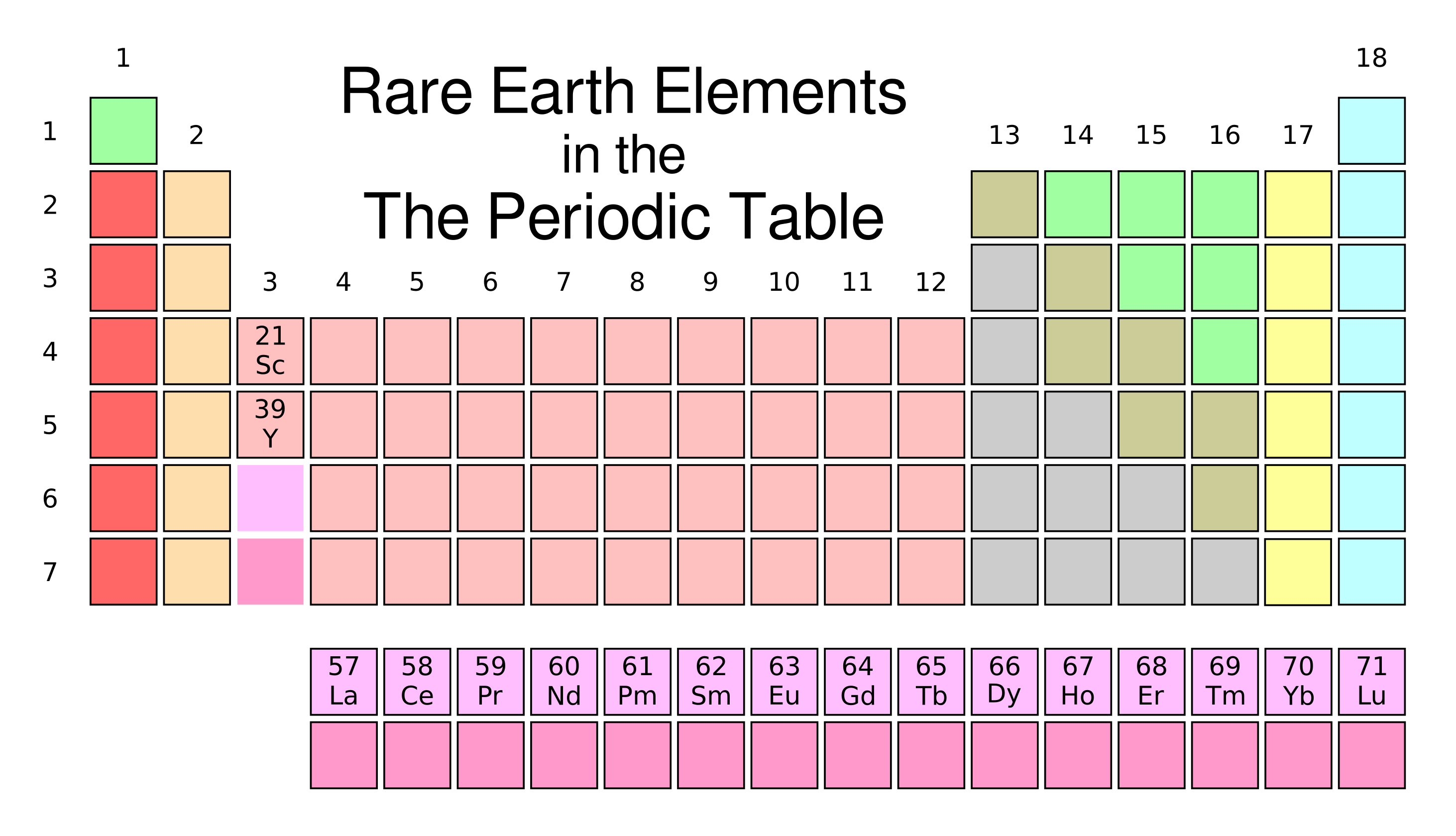
2022 Found on the periodic table as two separate rows, they are used in high-tech consumer products, including touch screens, electric vehicle motors, and computer hard drives. 2022 Here, there were lots of examples of storing aluminum as a chemical compound with elements below oxygen on the periodic table, such as sulfur or selenium. 2023 The company brands its four models of IEM (In-Ear Monitor) with symbols from the periodic table of elements. 117, tennessine, which now holds a place on the periodic table. 2016 In 2016, nuclear chemist Clarice Phelps was recognized as the first Black woman involved with the discovery of an element: No. 2021 In January, an international collaboration of scientists added four new elements to the periodic table: nihonium, moscovium, tennessine and oganesson. 2023 The histories hidden in the periodic table. Recent Examples on the Web Of all the elements in the periodic table, silicon has the highest capacity for combining with lithium.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
